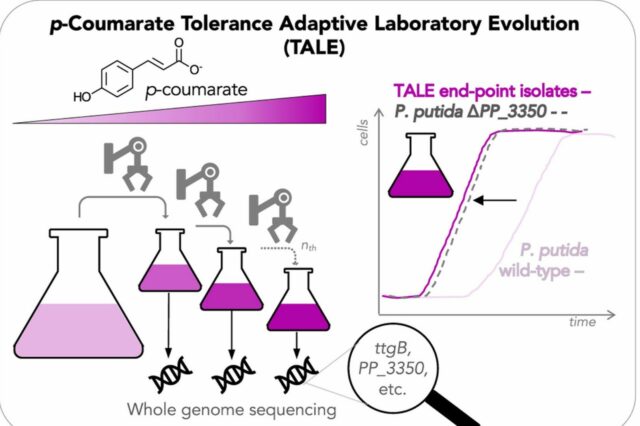
Adaptive laboratory evolution experiments were used to increase the tolerance of successive generations of bacteria. Credit: Mohamed, E. et. al., Metabolic Engineering Communications. Excerpt of the original, used under CC by 4.0*
Scientists collaborating through the Center for Bioenergy Innovation recently learned to use laboratory evolution and genetic engineering to develop a more robust strain of bacteria, better able to produce valuable chemicals out of hydroxycinnamic acids derived from lignin in plants.
Lignin gives structure and strength to plant cell walls and is difficult to break down when making biofuel or other products from plant feedstock, like poplar and switchgrass. Other scientists are developing efficient ways for microbes to break down lignin into smaller parts. However, those smaller parts are aromatic acids, which are hard for bacteria to tolerate. That challenge must be addressed before the bacteria can be used to funnel high concentrations of diverse lignin components into a single, valuable chemical product.
“These aromatic acids are released from the lignin and cause the bacteria cell quite a bit of stress,” said Allison Werner, a lead researcher on the recent study. “The purpose of this paper was to understand how to leverage evolution to identify ways to improve the cell’s robustness in the presence of these aromatic acids: to make cells grow faster and stronger, even with all these toxic compounds bumping against them.”
Research partners from the Lawrence Berkeley National Laboratory, the University of California, the National Renewable Energy Laboratory, the Joint BioEnergy Institute and the Technical University of Denmark each played key roles in the study coordinated by CBI.
First, bacteria were evolved in the lab, exposing them to progressively higher concentrations of aromatic acids. The bacterial cells that emerged were run through genome sequencing to better understand the mutations that allowed them to tolerate the high concentrations. The bacteria that were most successful had mutated a particular gene that, until this study, was understood generally to encode some sort of protein. CBI collaborators used computational modeling to predict that this gene encodes a specific protein called a porin, which forms channels through cell membranes. The authors hypothesized that the porin normally allowed the aromatic acids to rapidly enter a cell and toxically accumulate there. But in the mutated strain of bacteria, the aromatic acids could only passively cross the outer cell membrane, Werner said.
Using reverse engineering, scientists returned to the original bacteria and deleted the gene responsible for forming the porin. The genetically engineered bacteria achieved the same positive results as in the evolved bacteria. Without the porin, the bacteria grew slightly faster. More significantly, the lag phase – the period when bacteria are adjusting to their environment and unable to process the lignin components – was reduced by a factor of four, from 50 hours to 12, Werner said. That could significantly hasten the process of making valuable biofuel byproducts.
Their results demonstrated the effectiveness of using adaptive laboratory evolution in combination with genome sequencing and metabolic engineering.
Werner, a staff scientist at the National Renewable Energy Laboratory and a CBI Early Career Fellow, was a lead author in another recent study through a parallel track of CBI research. It focuses on developing a biocatalyst that could convert all three types of lignin into a single product, so less lignin would remain unused and unprofitable.
The goal is to ultimately combine the performance enhancements refined during both studies into a single bacterium, Werner said. “We’ve developed a way to make a strain more tolerant of the aromatic acids, and we’ve developed a strain that can convert more lignin,” she said. “We need to merge those into the same bacterium so that we have a robust and high-conversion biocatalyst.” Making lignin into desirable products, instead of viewing it as a waste stream from biorefineries, is a necessary step to make biofuel production more affordable and sustainable.
The Center for Bioenergy Innovation at ORNL is one of four DOE Bioenergy Research Centers focused on advancing biofuels and bioproducts for a vibrant domestic bioeconomy. CBI is accelerating the development of bioenergy-relevant plants and microbes to enable production of drop-in sustainable aviation fuel, bioproducts that sequester carbon indefinitely, and sustainable replacements for plastics and other environmentally harmful products. CBI research is supported by the Biological and Environmental Research program in DOE’s Office of Science.
*Image credit: Mohamed, E. et. al., Metabolic Engineering Communications. Excerpt of the original, used under CC by 4.0
